It is some three or four years since I had the honour of lecturing here one Friday evening upon the densities of oxygen and hydrogen gases, and upon the conclusions that might be drawn from the results. It is not necessary, therefore, that I should trouble you to-night with any detail as to the method by which gases can be accurately weighed. I must take that as known, merely mentioning that it is substantially the same as is used by all investigators nowadays, and introduced more than fifty years ago by Regnault. It was not until after that lecture that I turned my attention to nitrogen; and in the first instance I employed a method of preparing the gas which originated with Mr Vernon Harcourt, of Oxford. In this method the oxygen of ordinary atmospheric air is got rid of with the aid of ammonia. Air is bubbled through liquid ammonia, and then passed through a red-hot tube. In its passage the oxygen of the air combines with the hydrogen of the ammonia, all the oxygen being in that way burnt up and converted into water. The excess of ammonia is subsequently absorbed with acid, and the water by ordinary desiccating agents. That method is very convenient; and, when I had obtained a few concordant results by means of it, I thought that the work was complete, and that the weight of nitrogen was satisfactorily determined. But then I reflected that it is always advisable to employ more than one method, and that the method I had used--Mr Vernon Harcourt's method--was not that which had been used by any of those who had preceded me in weighing nitrogen. The usual method consists in absorbing the oxygen of air by means of red-hot copper; and I thought that I ought at least to give that method a trial, fully expecting to obtain forthwith a value in harmony with that already afforded by the ammonia method. The result, however, proved otherwise. The gas obtained by the copper method, as I may call it, proved to be one-thousandth part heavier than that obtained by the ammonia method; and, on repetition, that difference was only brought out more clearly. This was about three years ago. In order, if possible, to get further light upon a discrepancy which puzzled me very much, and which, at that time, I regarded only with disgust and impatience, I published a letter in Nature inviting criticisms from chemists who might be interested in such questions. I obtained various useful suggestions, but none going to the root of the matter. Several persons who wrote to me privately were inclined to think that the explanation was to be sought in a partial dissociation of the nitrogen derived from ammonia. For, before going further, I ought to explain that, in the nitrogen obtained by the ammonia method, some--about a seventh part--is derived from the ammonia, the larger part, however, being derived as usual from the atmosphere. If the chemically derived nitrogen were partly dissociated into its component atoms, then the lightness of the gas so prepared would be explained.
The next step in the enquiry was, if possible, to exaggerate the discrepancy. One's instinct at first is to try to get rid of a discrepancy, but I believe that experience shows such an endeavour to be a mistake. What one ought to do is to magnify a small discrepancy with a view to finding out the explanation; and, as it appeared in the present case that the root of the discrepancy lay in the fact that part of the nitrogen prepared by the ammonia method was nitrogen out of ammonia, although the greater part remained of common origin in both cases, the application of the principle suggested a trial of the weight of nitrogen obtained wholly from ammonia. This could easily be done by substituting pure oxygen for atmospheric air in the ammonia method, so that the whole, instead of only a part, of the nitrogen collected should be derived from the ammonia itself. The discrepancy was at once magnified some five times. The nitrogen so obtained from ammonia proved to be about one-half per cent. lighter than nitrogen obtained in the ordinary way from the atmosphere, and which I may call for brevity "atmospheric" nitrogen.
That result stood out pretty sharply from the first; but it was necessary to confirm it by comparison with nitrogen chemically derived in other ways. The table before you gives a summary of such results, the numbers being the weights in grams actually contained under standard conditions in the globe employed.
| By hot copper (1892) | 2.3103 |
| By hot iron (1893) | 2.3100 |
| By ferrous hydrate (1894) | 2.3102 |
| mean | 2.3102 |
| From nitric oxide | 2.3001 |
| From nitrous oxide | 2.2990 |
| From ammonium nitrite purified at a red heat | 2.2987 |
| From urea | 2.2985 |
| From ammonium nitrite purified in the cold | 2.2987 |
| mean | 2.2990 |
The difference is about 11 milligrams, or about one-half per cent.; and it was sufficient to prove conclusively that the two kinds of nitrogen--the chemically derived nitrogen and the atmospheric nitrogen--differed in weight, and therefore, of course, in quality, for some reason hitherto unknown.
I need not spend time in explaining the various precautions that were necessary in order to establish surely that conclusion. One had to be on one's guard against impurities, especially against the presence of hydrogen, which might seriously lighten any gas in which it was contained. I believe, however, that the precautions taken were sufficient to exclude all questions of that sort, and the result, which I published about this time last year, stood sharply out, that the nitrogen obtained from chemical sources was different from the nitrogen obtained from the air.
Well, that difference, admitting it to be established, was sufficient to show that some hitherto unknown gas is involved in the matter. It might be that the new gas was dissociated nitrogen, contained in that which was too light, the chemical nitrogen--and at first that was the explanation to which I leaned; but certain experiments went a long way to discourage such a supposition. In the first place, chemical evidence--and in this matter I am greatly dependent upon the kindness of chemical friends--tends to show that, even if ordinary nitrogen could be dissociated at all into its component atoms, such atoms would not be likely to enjoy any very long continued existence. Even ozone goes slowly back to the more normal state of oxygen; and it was thought that dissociated nitrogen would have even a greater tendency to revert to the normal condition. The experiment suggested by that remark was as follows--to keep chemical nitrogen--the too light nitrogen which might be supposed to contain dissociated molecules--for a good while, and to examine whether it changed in density. Of course it would be useless to shut up gas in a globe and weigh it, and then, after an interval, to weigh it again, for there would be no opportunity for any change of weight to occur, even although the gas within the globe had undergone some chemical alteration. It is necessary to re-establish the standard conditions of temperature and pressure which are always understood when we speak of filling a globe with gas, for I need hardly say that filling a globe with gas is but a figure of speech. Everything depends upon the temperature and pressure at which you work. However, that obvious point being borne in mind, it was proved by experiment that the gas did not change in weight by standing for eight months--a result tending to show that the abnormal lightness was not the consequence of dissociation.
Further experiments were tried upon the action of the silent electric discharge--both upon the atmospheric nitrogen and upon the chemically derived nitrogen--but neither of them seemed to be sensibly affected by such treatment; so that, altogether, the balance of evidence seemed to incline against the hypothesis of abnormal lightness in the chemically derived nitrogen being due to dissociation, and to suggest strongly, as almost the only possible alternative, that there must be in the atmospheric nitrogen some constituent heavier than true nitrogen.
At that point the question arose, What was the evidence that all the so-called nitrogen of the atmosphere was of one quality? And I remember--I think it was about this time last year, or a little earlier--putting the question to my colleague, Professor Dewar. His answer was that he doubted whether anything material had been done upon the matter since the time of Cavendish, and that I had better refer to Cavendish's original paper. That advice I quickly followed, and I was rather surprised to find that Cavendish had himself put this question quite as sharply as I could put it. Translated from the old-fashioned phraseology connected with the theory of phlogiston, his question was whether the inert ingredient of the air is really all of one kind; whether all the nitrogen of the air is really the same as the nitrogen of nitre. Cavendish not only asked himself this question, but he endeavoured to answer it by an appeal to experiment.
I should like to show you Cavendish's experiment in something like its original form. He inverted a U-tube filled with mercury, the legs standing in two separate mercury cups. He then passed up, so as to stand above the mercury, a mixture of nitrogen, or of air, and oxygen; and he caused an electric current from a frictional electrical machine like the one I have before me to pass from the mercury in the one leg to the mercury in the other, giving sparks across the intervening column of air. I do not propose to use a frictional machine to-night, but I will substitute for it one giving electricity of the same quality of the construction introduced by Mr Wimshurst, of which we have a fine specimen in the Institution. It stands just outside the door of the theatre, and will supply an electric current along insulated wires, leading to the mercury cups; and, if we are successful, we shall cause sparks to pass through the small length of air included above the columns of mercury. There they are; and after a little time you will notice that the mercury rises, indicating that the gas is sensibly absorbed under the influence of the sparks and of a piece of potash floating on the mercury. It was by that means that Cavendish established his great discovery of the nature of the inert ingredient in the atmosphere, which we now call nitrogen; and, as I have said, Cavendish himself proposed the question, as distinctly as we can do, Is this inert ingredient all of one kind? and he proceeded to test that question. He found, after days and weeks of protracted experiment, that, for the most part, the nitrogen of the atmosphere was absorbed in this manner, and converted into nitrous acid; but that there was a small residue remaining after prolonged treatment with sparks, and a final absorption of the residual oxygen. That residue amounted to about 1/120 part of the nitrogen taken; and Cavendish draws the conclusion that, if there be more than one inert ingredient in the atmosphere, at any rate the second ingredient is not contained to a greater extent than 1/120 part.
I must not wait too long over the experiment. Mr Gordon tells me that a certain amount of contraction has already occurred; and if we project the U upon the screen, we shall be able to verify the fact. It is only a question of time for the greater part of the gas to be taken up, as we have proved by preliminary experiments.
In what I have to say from this point onwards, I must be understood as speaking as much on behalf of Professor Ramsay as for myself. At the first, the work which we did was to a certain extent independent. Afterwards we worked in concert, and all that we have published in our joint names must be regarded as being equally the work of both of us. But, of course, Professor Ramsay must not be held responsible for any chemical blunder into which I may stumble to-night.
By his work and by mine the heavier ingredient in atmospheric nitrogen which was the origin of the discrepancy in the densities has been isolated, and we have given it the name of "argon". For this purpose we may use the original method of Cavendish, with the advantage of modern appliances. We can procure more powerful electric sparks than any which Cavendish could command by the use of the ordinary Ruhmkorff coil stimulated by a battery of Grove cells; and it is possible so to obtain evidence of the existence of argon. The oxidation of nitrogen by that method goes on pretty quickly. If you put some ordinary air, or better still, a mixture of air and oxygen, in a tube in which electric sparks are made to pass for a certain time, then in looking through the tube, you observe the well-known reddish-orange fumes of the oxides of nitrogen. I will not take up time in going through the experiment, but will merely exhibit a tube already prepared (image on screen).
One can work more efficiently by employing the alternate currents from dynamo machines which are now at our command. In this Institution we have the advantage of a public supply; and if I pass alternate currents originating in Deptford through this Ruhmkorff coil, which acts as what is now called a "high potential transformer", and allow sparks from the secondary to pass in an inverted test tube between platinum points, we shall be able to show in a comparatively short time a pretty rapid absorption of the gases. The electric current is led into the working chamber through bent glass tubes containing mercury, and provided at their inner extremities with platinum points. In this arrangement we avoid the risk, which would otherwise be serious, of a fracture just when we least desired it. I now start the sparks by switching on the Ruhmkorff to the alternate current supply; and, if you will take note of the level of the liquid representing the quantity of mixed gases included, I think you will see after, perhaps, a quarter of an hour that the liquid has very appreciably risen, owing to the union of the nitrogen and the oxygen gases under the influence of the electrical discharge, and subsequent absorption of the resulting compound by the alkaline liquid with which the gas space is enclosed.
By means of this little apparatus, which is very convenient for operations upon a moderate scale, such as analyses of "nitrogen" for the amount of argon that it may contain, we are able to get an absorption of about 80 cubic centimetres per hour, or about 4 inches along this test tube, when all is going well. In order, however, to effect the isolation of argon on any considerable scale by means of the oxygen method, we must employ an apparatus still more enlarged. The isolation of argon requires the removal of nitrogen, and, indeed, of very large quantities of nitrogen, for, as it appears, the proportion of argon contained in atmospheric nitrogen is only about one per cent., so that for every litre of argon that you wish to get you must eat up some hundred litres of nitrogen. That, however, can be done upon an adequate scale by calling to our aid the powerful electric discharge now obtainable by means of the alternate current supply and high potential transformers.
In what I have done upon this subject I have had the advantage of the advice of Mr Crookes, who some years ago drew special attention to the electric discharge or flame, and showed that many of its properties depended upon the fact that it had the power of causing, upon a very considerable scale, a combination of the nitrogen and the oxygen of the air in which it was made.
I had first thought of showing in the lecture room the actual apparatus which I have employed for the concentration of argon; but the difficulty is that, as the apparatus has to be used, the working parts are almost invisible, and I came to the conclusion that it would really be more instructive as well as more convenient to show the parts isolated, a very little effort of imagination being then all that is required in order to reconstruct in the mind the actual arrangements employed.
First, as to the electric arc or flame itself. We have here a transformer made by Pike and Harris. It is not the one that I have used in practice; but it is convenient for certain purposes, and it can be connected by means of a switch with the alternate currents of 100 volts furnished by the Supply Company. The platinum terminals that you see here are modelled exactly upon the plan of those which have been employed in practice. I may say a word or two on the question of mounting. The terminals require to be very massive on account of the heat evolved. In this case they consist of platinum wire doubled upon itself six times. The platinums are continued by iron wires going through glass tubes, and attached at the ends to the copper leads. For better security, the tubes themselves are stopped at the lower ends with corks and charged with water, the advantage being that, when the whole arrangement is fitted by means of an indiarubber stopper into a closed vessel, you have a witness that, as long as the water remains in position, no leak can have occurred through the insulating tubes conveying the electrodes.
Now, if we switch on the current and approximate the points sufficiently, we get the electric flame. There you have it. It is, at present, showing a certain amount of soda. That in time would burn off. After the arc has once been struck, the platinums can be separated; and then you have two tongues of fire ascending almost independently of one another, but meeting above. Under the influence of such a flame, the oxygen and the nitrogen of the air combine at a reasonable rate, and in this way the nitrogen is got rid of. It is now only a question of boxing up the gas in a closed space, where the argon concentrated by the combustion of the nitrogen can be collected. But there are difficulties to be encountered here. One cannot well use anything but a glass vessel. There is hardly any metal available that will withstand the action of strong alkali and of the nitrous fumes resulting from the flame. One is practically limited to glass. The glass vessel employed is a large flask with a single neck, about half full of caustic alkali. The electrodes are carried through the neck by means of an indiarubber bung provided also with tubes for leading in the gas. The electric flame is situated at a distance of only about half an inch above the caustic alkali. In that way an efficient circulation is established; the hot gases as they rise from the flame strike the top, and then as they come round again in the course of the circulation they pass sufficiently close to the caustic alkali to ensure an adequate removal of the nitrous fumes.
There is another point to be mentioned. It is necessary to keep the vessel cool; otherwise the heat would soon rise to such a point that there would be excessive generation of steam, and then the operation would come to a standstill. In order to meet this difficulty the upper part of the vessel is provided with a water-jacket, in which a circulation can be established. No doubt the glass is severely treated, but it seems to stand it in a fairly amiable manner.
By means of an arrangement of this kind, taking nearly three horse-power from the electric supply, it is possible to consume nitrogen at a reasonable rate. The transformers actually used are the "Hedgehog" transformers of Mr Swinburne, intended to transform from 100 to 2400 volts. By Mr Swinburne's advice I have used two such, the fine wires being in series so as to accumulate the electrical potential and the thick wires in parallel. The rate at which the mixed gases are absorbed is about seven litres per hour; and the apparatus, when once fairly started, works very well as a rule, going for many hours without attention. At times the arc has a trick of going out, and it then requires to be restarted by approximating the platinums. We have already worked 14 hours on end, and by the aid of one or two automatic appliances it would, I think, be possible to continue operation day and night.
The gases, air and oxygen in about equal proportions, are mixed in a large gas-holder, and are fed in automatically as required. The argon gradually accumulates; and when it is desired to stop operations the supply of nitrogen is cut off, and only pure oxygen allowed admittance. In this way the remaining nitrogen is consumed, so that, finally, the working vessel is charged with a mixture of argon and oxygen only, from which the oxygen is removed by ordinary well-known chemical methods. I may mention that at the close of the operation, when the nitrogen is all gone, the arc changes its appearance and becomes of a brilliant blue colour.
I have said enough about this method, and I must now pass on to the alternative method which has been very successful in Professor Ramsay's hands--that of absorbing nitrogen by means of red-hot magnesium. By the kindness of Professor Ramsay and Mr Matthews, his assistant, we have here the full scale apparatus before us almost exactly as they use it. On the left there is a reservoir of nitrogen derived from air by the simple removal of oxygen. The gas is then dried. Here it is bubbled through sulphuric acid. It then passes through a long tube made of hard glass and charged with magnesium in the form of thin turnings. During the passage of the gas over the magnesium at a bright red heat, the nitrogen is absorbed in a great degree, and the gas which finally passes through is immensely richer in argon than that which first enters the hot tube. At the present time you see a tolerably rapid bubbling on the left, indicative of the flow of atmospheric nitrogen into the combustion furnace; whereas, on the right, the outflow is very much slower. Care must be taken to prevent the heat rising to such a point as to soften the glass. The concentrated argon is collected in a second gas-holder, and afterwards submitted to further treatment. The apparatus employed by Professor Ramsay in the subsequent treatment is exhibited in the diagram, and is very effective for its purpose; but I am afraid that the details of it would not readily be followed from any explanation that I could give in the time at my disposal. The principle consists in the circulation of the mixture of nitrogen and argon over hot magnesium, the gas being made to pass round and round until the nitrogen is effectively removed from it. At the end that operation, as in the case of the oxygen method, proceeds somewhat slowly. When the greater part of the nitrogen is gone, the remainder seems to be unwilling to follow, and it requires somewhat protracted treatment in order to be sure that the nitrogen has wholly disappeared. When I say "wholly disappeared", that perhaps, would be too much to say in any case. What we can say is that the spectrum test is adequate to show the presence, or at any rate to show the addition, of about one-and-a-half per cent. of nitrogen to argon as pure as we can get it; so that it is fair to argue that any nitrogen at that stage remaining in the argon is only a small fraction of one-and-a-half per cent.
I should have liked at this point to be able to give advice as to which of the two methods--the oxygen method or the magnesium method--is the easier and the more to be recommended; but I confess that I am quite at a loss to do so. One difficulty in the comparison arises from the fact that they have been in different hands. As far as I can estimate, the quantities of nitrogen eaten up in a given time are not very different. In that respect, perhaps, the magnesium method has some advantage; but, on the other hand, it may be said that the magnesium process requires a much closer supervision, so that, perhaps, fourteen hours of the oxygen process may not unfairly compare with eight hours or so of the magnesium method. In practice a great deal would depend upon whether in any particular laboratory alternate currents are available from a public supply. If the alternate currents are at hand, I think it may probably be the case that the oxygen method is the easier; but, otherwise, the magnesium method would, probably, be preferred, especially by chemists who are familiar with operations conducted in red-hot tubes.
I have here another experiment illustrative of the reaction between magnesium and nitrogen. Two rods of that metal are suitably mounted in an atmosphere of nitrogen, so arranged that we can bring them into contact and cause an electric arc to form between them. Under the action of the heat of the electric arc the nitrogen will combine with the magnesium; and if we had time to carry out the experiment we could demonstrate a rapid absorption of nitrogen by this method. When the experiment was first tried, I had hoped that it might be possible, by the aid of electricity, to start the action so effectively that the magnesium would continue to burn independently under its own developed heat in the atmosphere of nitrogen. Possibly, on a larger scale, something of this sort might succeed, but I bring it forward here only as an illustration. We turn on the electric current, and bring the magnesiums together. You see a brilliant green light, indicating the vaporisation of the magnesium. Under the influence of heat the magnesium burns, and there is collected in the glass vessel a certain amount of brownish-looking powder which consists mainly of the nitride of magnesium. Of course, if there is any oxygen present it has the preference, and the ordinary white oxide of magnesium is formed.
The gas thus isolated is proved to be inert by the very fact of its isolation. It refuses to combine under any circumstances in which nitrogen, itself always considered very inert, does combine--both in the case of the oxygen treatment and in the case of the magnesium treatment; and these facts are, perhaps, almost enough to justify the name which we have suggested for it. But in addition to this, it has been proved to be inert under a considerable variety of other conditions such as might have been expected to tempt it into combination. I will not recapitulate all the experiments which have been tried, almost entirely by Professor Ramsay, to induce the gas to combine. Hitherto, in our hands, it has not done so; and I may mention that recently, since the publication of the abstract of our paper read before the Royal Society, argon has been submitted to the action of titanium at red heat, titanium being a metal having a great affinity for nitrogen, and that argon has resisted the temptation to which nitrogen succumbs. We never have asserted, and we do not now assert, that argon can under no circumstances be got to combine. That would, indeed, be a rash assertion for any one to venture upon; and only within the last few weeks there has been a most interesting announcement by M. Berthelot, of Paris, that, under the action of the silent electric discharge, argon can be absorbed when treated in contact with the vapour of benzine. Such a statement, coming from so great an authority, commands our attention; and if we accept the conclusion, as I suppose we must do, it will follow that argon has, under those circumstances, combined.
Argon is rather freely soluble in water. That is a thing that troubled us at first in trying to isolate the gas; because, when one was dealing with very small quantities, it seemed to be always disappearing. In trying to accumulate it we made no progress. After a sufficient quantity had been prepared, special experiments were made on the solubility of argon in water. It has been found that argon, prepared both by the magnesium method and by the oxygen method, has about the same solubility in water as oxygen--some two-and-a half times the solubility of nitrogen. This suggests, what has been verified by experiment, that the dissolved gases of water should contain a larger proportion or argon than does atmospheric nitrogen. I have here an apparatus of a somewhat rough description, which I have employed in experiments of this kind. The boiler employed consists of an old oil-can. The water is supplied to it and drawn from it by coaxial tubes of metal. The incoming cold water flows through the outer annulus between the two tubes. The outgoing hot water passes through the inner tube, which ends in the interior of the vessel at a higher level. By means of this arrangement the heat of the water which has done its work is passed on to the incoming water not yet in operation, and in that way a limited amount of heat is made to bring up to the boil a very much larger quantity of water than would otherwise be possible, the greater part of the dissolved gases being liberated at the same time. These are collected in the ordinary way. What you see in this flask is dissolved air collected out of water in the course of the last three or four hours. Such gas, when treated as if it were atmospheric nitrogen, that is to say after removal of the oxygen and minor impurities, is found to be decidedly heavier than atmospheric nitrogen to such an extent as to indicate that the proportion of argon contained is about double. It is obvious, therefore, that the dissolved gases of water form a convenient source of argon, by which some of the labor of separation from air is obviated. During the last few weeks I have been supplied from Manchester by Mr Macdougall, who has interested himself in this matter, with a quantity of dissolved gases obtained from the condensing water of his steam engine.
As to the spectrum, we have been indebted from the first to Mr Crookes, and he has been good enough to-night to bring some tubes which he will operate, and which will show you at all events the light of the electric discharge in argon. I cannot show you the spectrum of argon, for unfortunately the amount of light from a vacuum tube is not sufficient for the projection of its spectrum. Under some circumstances the light is red, and under other circumstances it is blue. Of course when these lights are examined with the spectroscope--and they have been examined by Mr Crookes with great care--the differences in the colour of the light translate themselves into different groups of spectrum lines. We have before us Mr Crookes' map, showing the two spectra upon a very large scale. The upper is the spectrum of the blue light; the lower is the spectrum of the red light; and it will be seen that they differ very greatly. Some lines are common to both; but a great many lines are seen only in the red, and others are seen only in the blue. It is astonishing to notice what trifling changes in the conditions of the discharge bring about such extensive alterations in the spectrum.
One question of great importance upon which the spectrum throws light is, Is the argon derived from the oxygen method really the same as the argon derived by the magnesium method? By Mr Crookes' kindness I have had an opportunity of examining the spectra of the two gases side by side, and such examination as I could make revealed no difference whatever in the two spectra, from which, I suppose, we may conclude either that the gases are absolutely the same, or, if they are not the same, that at any rate the ingredients by which they differ cannot be present in more than a small proportion in either of them.
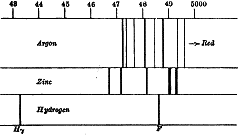
My own observations upon the spectrum have been made principally at atmospheric pressure. In the ordinary process of sparking, the pressure is atmospheric; and, if we wish to look at the spectrum, we have nothing more to do than to include a jar in the circuit, and to put a direct-vision prism to the eye. At my request, Professor Schuster examined some tubes containing argon at atmospheric pressure prepared by the oxygen method, and I have here the diagram of a characteristic group. He also placed upon the sketch some of the lines of zinc, which were very convenient as directing one exactly where to look. See figure.

Within the last few days, Mr Crookes has charged a radiometer with argon. When held in the light from the electric lamp, the vanes revolve rapidly. Argon is anomalous in many respects, but not, you see, in this.
Next, as to the density of argon. Professor Ramsay has made numerous and careful observations upon the density of the gas prepared by the magnesium method, and he finds a density of about 19.9 as compared with hydrogen. Equally satisfactory observations upon the gas derived by the oxygen method have not yet been made, but there is no reason to suppose that the density is different, such numbers as 19.7 having been obtained.
One of the most interesting matters in connection with argon, however, is what is known as the ratio of the specific heats. I must not stay to elaborate the questions involved, but it will be known to many who hear me that the velocity of sound in a gas depends upon the ratio of two specific heats--the specific heat of a gas measured at constant pressure, and the specific heat measured at constant volume. If we know the density of a gas, and also the velocity of sound in it, we are in a position to infer this ratio of specific heats; and by means of this method, Professor Ramsay has determined the ratio in the case of argon, arriving at the very remarkable result that the ratio of specific heats is represented by the number 1.65, approaching very closely to the theoretical limit, 1.67. The number 1.67 would indicate that the gas has no energy except energy of translation of its molecules. If there is any other energy than that, it would show itself by this number dropping below 1.67. Ordinary gases, oxygen, nitrogen, hydrogen, &c., do drop below, giving the number 1.4. Other gases drop lower still. If the ratio of specific heats is 1.65, practically 1.67, we may infer that the whole energy of motion is translational; and from that it would seem to follow by arguments which, however, I must not stop to elaborate, that the gas must be of the kind called by chemists monatomic.
I had intended to say something of the operation of determining the ratio of specific heats, but time will not allow. The result is, no doubt, very awkward. Indeed, I have seen some indications that the anomalous properties of argon are brought as a kind of accusation against us. But we had the very best intentions in the matter. The facts were too much for us; and all that we can do now is to apologise for ourselves and for the gas.
Several questions may be asked, upon which I should like to say a word or two, if you will allow me to detain you a little longer. The first question (I do not know whether I need ask it) is, Have we got hold of a new gas at all? I had thought that that might be passed over, but only this morning I read in a technical journal the suggestion that argon was our old friend nitrous oxide. Nitrous oxide has roughly the density of argon; but that, so far as I can see, is the only point of resemblance between them.
Well, supposing that there is a new gas, which I will not stop to discuss, because I think that the spectrum alone would be enough to prove it, the next question that may be asked is, Is it in the atmosphere? This matter naturally engaged our earnest attention at an early stage of the enquiry. I will only indicate in a few words the arguments which seem to us to show that the answer must be in the affirmative.
In the first place, if argon be not in the atmosphere, the original discrepancy of densities which formed the starting-point of the investigation remains unexplained, and the discovery of the new gas has been made upon a false clue. Passing over that, we have the evidence from the blank experiments, in which nitrogen originally derived from chemical sources is treated either with oxygen or with magnesium, exactly as atmospheric nitrogen is treated. If we use atmospheric nitrogen, we get a certain proportion of argon, about 1 per cent. If we treat chemical nitrogen in the same way we get, I will not say absolutely nothing, but a mere fraction of what we should get had atmospheric nitrogen been the subject. You may ask, Why do we get any fraction at all from chemical nitrogen? It is not difficult to explain the small residue, because in the manipulation of the gases large quantities of water are used; and, as I have already explained, water dissolves argon somewhat freely. In the processes of manipulation some of the argon will come out of solution and it remains after all the nitrogen has been consumed.
Another wholly distinct argument is founded upon the method of diffusion introduced by Graham. Graham showed that if you pass gas along porous tubes you alter the composition, if the gas is a mixture. The lighter constituents go more readily through the pores than do the heavier ones. The experiment takes this form. A number of tobacco pipes--eight in the actual arrangement--are joined together in series with indiarubber junctions, and they are put in a space in which a vacuum can be made, so that the space outside the porous pipes is vacuous or approximately so. Through the pipes ordinary air is led. One end may be regarded as open to the atmosphere. The other end is connected with an aspirator so arranged that the gas collected is only some 2 per cent. of that which leaks through the porosities. The case is like that of an Australian river drying up almost to nothing in the course of its flow. Well, if we treat air in that way, collecting only the small residue which is less willing than the remainder to penetrate the porous walls, and then prepare "nitrogen" from it by removal of oxygen and moisture, we obtain a gas heavier than atmospheric nitrogen, a result which proves that the ordinary nitrogen of the atmosphere is not a simple body, but is capable of being divided into parts by so simple an agent as the tobacco pipe.
If it be admitted that the gas is in the atmosphere, the further question arises as to its nature.
At this point I would wish to say a word of explanation. Neither in our original announcement at Oxford, nor at any time since, until the 31st of January, did we utter a word suggesting that argon was an element; and it was only after the experiments upon the specific heats that we thought that we had sufficient to go upon in order to make any such suggestion in public. I will not insist that the observation is absolutely conclusive. It is certainly strong evidence. But the subject is difficult, and one that has given rise to some difference of opinion among physicists. At any rate this property distinguishes argon very sharply from all the ordinary gases.
One question which occurred to us at the earliest stage of the enquiry, as soon as we knew that the density was not very different from 21, was the question of whether, possibly, argon could be a more condensed form of nitrogen, denoted chemically by the symbol N3. There seem to be several difficulties in the way of this supposition. Would such a constitution be consistent with the ratio of specific heats (1.65)? That seems extremely doubtful. Another question is, Can the density really be as high as 21, the number required on the supposition of N3? As to this matter, Professor Ramsay has repeated his measurements of density, and he finds that he cannot get even so high as 20. To suppose that the density of argon is really 21, and that it appears to be 20 in consequence of nitrogen still mixed with it, would be to suppose a contamination with nitrogen out of all proportion to what is probable. It would mean some 14 per cent. of nitrogen, whereas it seems that from one-and-a-half to two per cent. is easily enough detected by the spectroscope. Another question that may be asked is, Would N3 require so much cooling to condense it as argon requires?
There is one other matter on which I would like to say a word--the question as to what N3 would be like if we had it. There seems to be a great discrepancy of opinions. Some high authorities, among whom must be included, I see, the celebrated Mendeleef, consider that N3 would be an exceptionally stable body; but most of the chemists with whom I have consulted are of the opinion that N3 would be explosive, or, at any rate, absolutely unstable. That is a question which may be left for the future to decide. We must not attempt to put these matters too positively. The balance of evidence still seems to be against the supposition that argon is N3, but for my part I do not wish to dogmatise.
A few weeks ago we had an eloquent lecture from Professor Rücker on the life and work of the illustrious Helmholtz. It will be known to many that during the last few months of his life Helmholtz lay prostrate in a semi-paralysed condition, forgetful of many things, but still retaining a keen interest in science. Some little while after his death we had a letter from his widow, in which she described how interested he had been in our preliminary announcement at Oxford upon this subject, and how he desired the account of it to be read to him over again. He added the remark, "I always thought that there must be something more in the atmosphere."
 Back to the list of selected historical papers.
Back to the list of selected historical papers. Back to the top of Classic Chemistry.
Back to the top of Classic Chemistry.