John Dalton (1766-1844)
[excerpts], (Manchester, 1808) [from facsimile edition (London: Dawson)]
CHAP. II. On the Constitution of Bodies.
There are three distinctions in the kinds of bodies, or three states, which have more especially claimed the attention of philosophical chemists; namely, those which are marked by the terms elastic fluids, liquids, and solids. A very famous instance is exhibited to us in water, of a body, which, in certain circumstances, is capable of assuming all the three states. In steam we recognise a perfectly elastic fluid, in water a perfect liquid, and in ice a complete solid. These observations have tacitly led to the conclusion which seems universally adopted, that all bodies of sensible magnitude, whether liquid or solid, are constituted of a vast number of extremely small particles, or atoms of matter bound together by a force of attraction, which is more or less powerful according to circumstances, and which as it endeavours to prevent their separation, is very properly called in that view, attraction of cohesion; but as it collects them from a dispersed state (as from steam into water) it is called, attraction of aggregation, or more simply affinity. Whatever names it may go by, they still signify one and the same power. It is not my design to call in question this conclusion, which appears completely satisfactory; but to shew that we have hitherto made no use of it, and that the consequence of the neglect, has been a very obscure view of chemical agency, which is daily growing more so in proportion to the new lights attempted to be thrown upon it.
The opinions I more particularly allude to, are those of Berthollet on the Laws of chemical affinity; such as that chemical affinity is proportional to the mass, and that in all chemical unions, there exist insensible gradations in the proportions of the constituent principles. The inconsistence of these opinions, both with reason and observation, cannot, I think, fail to strike every one who takes a proper view of the phenomena.
Whether the ultimate particles of a body, such as water, are all alike, that is, of the same figure, weight, &c. is a question of some importance. From what is known, we have no reason to apprehend a diversity in the particulars: if it does exist in water, it must equally exist in the elements constituting water, namely, hydrogen and oxygen. Now it is scarcely possible to conceive how the aggregates of dissimilar particles should be so uniformly the same. If some of the particles of water were heavier than others, if a parcel of the liquid on any occasion were constituted principally of these heavier particles, it must be supposed to affect the specific gravity of the mass, a circumstance not known. Similar observations may be made on other substances. Therefore we may conclude that the ultimate particles of all homogeneous bodies are perfectly alike in weight, figure, &c. In other words, every particle of water is like every other particle of water; every particle of hydrogen is like every other particle of hydrogen, &c.
Besides the force of attraction, which, in one character or another, belongs universally to ponderable bodies, we find another force that is likewise universal, or acts upon all matter which comes under our cognisance, namely, a force of repulsion. This is now generally, and I think properly, ascribed to the agency of heat. An atmosphere of this subtile fluid constantly surrounds the atoms of all bodies, and prevents them from being drawn into actual contact. This appears to be satisfactorily proved by the observation, that the bulk of a body may be diminished by abstracting some of its heat: But from what has been stated in the last section, it should seem that enlargement and diminution of bulk depend perhaps more on the arrangement, than on the size of the ultimate particles. Be this as it may, we cannot avoid inferring from the preceding doctrine on heat, and particularly from the section on the natural zero of temperature, that solid bodies, such as ice, contain a large portion, perhaps 4/5 of the heat which the same are found to contain in an elastic state, as steam.
We are now to consider how these two great antagonist powers of attraction and repulsion are adjusted, so as to allow of the three different states of elastic fluids, liquids, and solids. We shall divide the subject into four Sections; namely, first, on the constitution of pure elastic fluids; second, on the constitution of mixed elastic fluids; third, on the constitution of liquids, and fourth, on the constitution of solids.
[I have omitted the sections of chapter II. --CJG]
CHAP. III. On Chemical Synthesis.
When any body exists in the elastic state, its ultimate particles are separated from each other to a much greater distance than in any other state; each particle occupies the centre of a comparatively large sphere, and supports its dignity by keeping all the rest, which by their gravity, or otherwise are disposed to encroach upon it, at a respectful distance. When we attempt to conceive the number of particles in an atmosphere, it is somewhat like attempting to conceive the number of stars in the universe; we are confounded with the thought. But if we limit the subject, by taking a given volume of any gas, we seem persuaded that, let the divisions be ever so minute, the number of particles must be finite; just as in a given space of the universe, the number of stars and planets cannot be infinite.
Chemical analysis and synthesis go no farther than to the separation of particles one from another, and to their reunion. No new creation or destruction of matter is within the reach of chemical agency. We might as well attempt to introduce a new planet into the solar system, or to annihilate one already in existence, as to create or destroy a particle of hydrogen. All the changes we can produce, consist in separating particles that are in a state of cohesion or combination, and joining those that were previously at a distance.
In all chemical investigations, it has justly been considered an important object to ascertain the relative weights of the simples which constitute a compound. But unfortunately the enquiry has terminated here; whereas from the relative weights in the mass, the relative weights of the ultimate particles or atoms of the bodies might have been inferred, from which their number and weight in various other compounds would appear, in order to assist and to guide future investigations, and to correct their results. Now it is one great object of this work, to shew the importance and advantage of ascertaining the relative weights of the ultimate particles, both of simple and compound bodies, the number of simple elementary particles which constitute one compound particle, and the number of less compound particles which enter into the formation of one more compound particle.
If there are two bodies, A and B, which are disposed to combine, the following is the order in which the combinations may take place, beginning with the most simple: namely,
- 1 atom of A + 1 atom of B = 1 atom of C, binary.
- 1 atom of A + 2 atoms of B = 1 atom of D, ternary.
- 2 atoms of A + 1 atom of B = 1 atom of E, ternary.
- 1 atom of A + 3 atoms of B = 1 atom of F, quarternary.
- 3 atoms of A + 1 atom of B = 1 atom of G, quarternary.
- &c. &c.
The following general rules may be adopted as guides in all our investigations respecting chemical synthesis.
- 1st. When only one combination of two bodies can be obtained, it must be presumed to be a binary one, unless some other cause appear to the contrary.
- 2d. When two combinations are observed, they must be presumed to be a binary and a ternary.
- 3d. When three combinations are observed, they must be presumed to be a binary, and the other two ternary.
- 4th. When four combinations are observed, we should expect one binary, two ternary, and one quarternary, &c.
- 5th. A binary compound should always be specifically heavier than the mere mixture of its two ingredients.
- 6th. A ternary compound should be specifically heavier than the mixture of a binary and a simple, which would, if combined, constitute it; &c.
- 7th. The above rules and observations equally apply, when two bodies, such as C and D, D and E, &c. are combined.
From the application of these rules, to the chemical facts already well ascertained, we deduce the following conclusions; 1st. That water is a binary compound of hydrogen and oxygen, and the relative weights of the two elementary atoms are as 1:7, nearly; 2d. That ammonia is a binary compound of hydrogen and azote, and the relative weights of the two atoms are as 1:5, nearly; 3d. That nitrous gas is a binary compound of azote and oxygen, the atoms of which weigh 5 and 7 respectively; that nitric acid is a binary or ternary compound according as it is derived, and consists of one atom of azote and two of oxygen, together weighing 19; that nitrous oxide is a compound similar to nitric acid, and consists of one atom of oxygen and two of azote, weighing 17; that nitrous acid is a binary compound of nitric acid and nitrous gas, weighing 31; that oxynitric acid is a binary compound of nitric acid with oxygen, weighing 26; 4th. That carbonic oxide is a binary compound, consisting of one atom of charcoal, and one of oxygen, together weighing nearly 12; that carbonic acid is a ternary compound, (but sometimes binary) consisting of one atom of charcoal, and two of oxygen, weighing 19; &c. &c. In all these cases the weights are expressed in atoms of hydrogen, each of which is denoted by unity.
In the sequel, the facts and experiments from which these conclusions are derived, will be detailed; as well as a great variety of others from which are inferred the constitution and weight of the ultimate particles of the principal acids, the alkalis, the earths, the metals, the metallic oxides and sulphurets, the long train of neutral salts, and in short, all the chemical compounds which have hitherto obtained a tolerably good analysis. Several of the conclusions will be supported by original experiments.
From the novelty as well as importance of the ideas suggested in this chapter, it is deemed expedient to give plates, exhibiting the mode of combination in some of the more simple cases. A specimen of these accompanies this first part. The elements or atoms of such bodies as are conceived at present to be simple, are denoted by a small circle, with some distinctive mark; and the combinations consist in the juxta-position of two or more of these; when three or more particles of elastic fluids are combined together in one, it is supposed that the particles of the same kind repel each other, and therefore take their stations accordingly.
END OF PART THE FIRST.
TABLE OF ATOMIC WEIGHTS
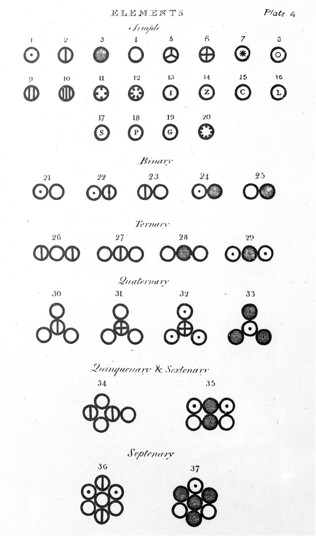
|
| 1. | Hydrogen, its relative weight | 1
| | 2. | Azote | 5
| | 3. | Carbone or charcoal | 5
| | 4. | Oxygen | 7
| | 5. | Phosphorous | 9
| | 6. | Sulphur | 13
| | 7. | Magnesia | 20
| | 8. | Lime | 23
| | 9. | Soda | 28
| | 10. | Potash | 42
| | 11. | Strontites | 46
| | 12. | Barytes | 68
| | 13. | Iron | 38
| | 14. | Zinc | 56
| | 15. | Copper | 56
| | 16. | Lead | 95
| | 17. | Silver | 100
| | 18. | Platina | 100
| | 19. | Gold | 140
| | 20. | Mercury | 167 |
21. An atom of water or steam, composed of 1 of oxygen and 1 of hydrogen, retained in physical contact by a strong affinity, and supposed to be surrounded by a common atmosphere of heat; its relative weight = 8
| 22. | An atom of ammonia, composed of 1 of azote and 1 of hydrogen | 6
| | 23. | An atom of nitrous gas, composed of 1 of azote and 1 of oxygen | 12
| | 24. | An atom of olefiant gas, composed of 1 of carbone and 1 of hydrogen | 6
| | 25. | An atom of carbonic oxide composed of 1 of carbone and 1 of oxygen | 12
| | 26. | An atom of nitrous oxide, 2 azote + 1 oxygen | 17
| | 27. | An atom of nitric acid, 1 azote + 2 oxygen | 19
| | 28. | An atom of carbonic acid, 1 carbone + 2 oxygen | 19
| | 29. | An atom of carburetted hydrogen, 1 carbone + 2 hydrogen | 7
| | 30. | An atom of oxynitric acid, 1 azote + 3 oxygen | 26
| | 31. | An atom of sulphuric acid, 1 sulphur + 3 oxygen | 34
| | 32. | An atom of sulphuretted hydrogen, 1 sulphur + 3 hydrogen | 16
| | 33. | An atom of alcohol, 3 carbone, + 1 hydrogen | 16
| | 34. | An atom of nitrous acid, 1 nitric acid + 1 nitrous gas | 31
| | 35. | An atom of acetous acid, 2 carbone + 2 water | 26
| | 36. | An atom of nitrate of ammonia, 1 nitric acid + 1 ammonia + 1 water | 33
| | 37. | An atom of sugar, 1 alcohol + 1 carbonic acid | 35 | |
 Back to the list of selected historical papers.
Back to the list of selected historical papers.
 Back to the top of Classic Chemistry.
Back to the top of Classic Chemistry.

 Back to the list of selected historical papers.
Back to the list of selected historical papers.